Your pain
matters
to us.
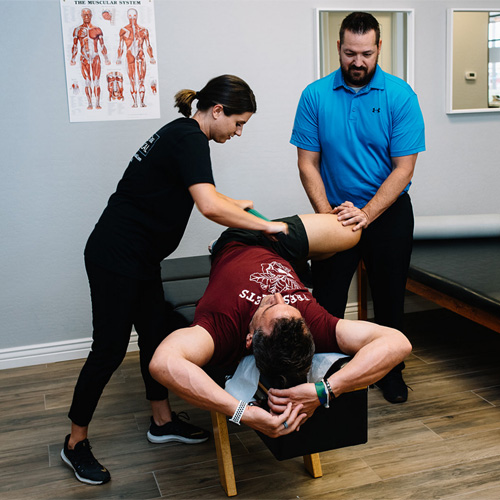
We know that one size does not fit all. We treat you as a whole person, never a symptom. Our patient’s success stems from working with each individual to meet their personal needs, never cookie-cutter service. We strive to listen to and understand the uniqueness of your conditions so we can give you the appropriate treatment and set you up for success.
STATE – OF – THE – ART CARE
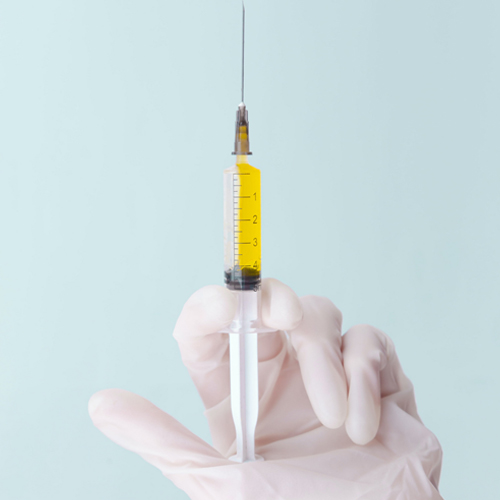
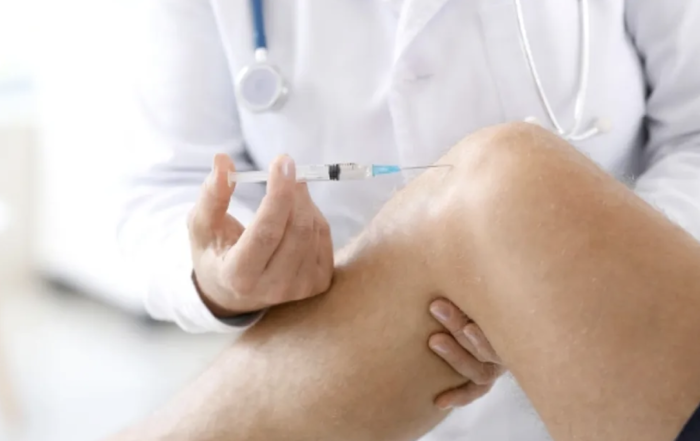
Trigger Point Injections
A trigger point injection (TPI), is an injection of an anti-inflammatory that is given directly into the trigger point for pain management.
E-Stim Therapy
E-stim uses electrical pulses to target muscles or nerves.. E-stim therapy for muscle recovery sends signals to targeted muscles to make them contract.
Invisa-RED
invisa-RED uses a combination of specific frequencies from both red and infrared laser light energy. The combination of dual coherent frequencies enables the light energy to penetrate deep into fat generating a significant response in cellular functions and stimulation of the body’s natural metabolism
Regenerative Stem-Cell Therapy
Regenerative stem cell therapy, promotes the repair response of diseased, dysfunctional or injured tissue using stem cells or their derivatives. …
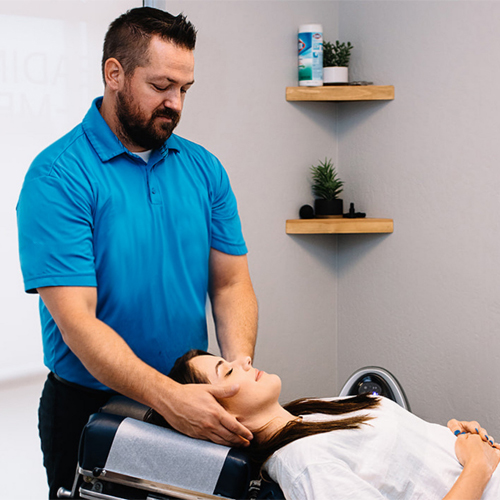
Spinal Decompression
Spinal decompression is a type of traction therapy applied to the spine in an attempt to bring about benefits including: Creating a negative intradiscal pressure to promote retraction or repositioning of the herniated or bulging disc material.
Who We Are
Our Mission
At Leading Edge Medical, our mission is to treat each patient with as much care as we would our own family. We believe in a team approach to give you the most complete care possible. We are passionate about your success and we don’t give up easily. Whatever ales you, we want to find you the proper treatment and get you on the road to a pain-free life. Along with our in-office staff, we work with doctors and surgeons to find the care that is right for you.
Meet the Team

READ OUR 5 STAR YELP REVIEWS





PRF for joint pain
Wound healing is a complex process where many cell types interact with one another in order to repair and regenerate damages tissues. While many regenerative agents currently exist in the market to help speed [...]
Could I Be Suffering From a Herniated Disc?
While we may not always know what is wrong with our body, we do know there’s something not quite right. If you’re experiencing low back pain that radiates, or if you’re feeling a pain, [...]
Is Your Teenager At Risk of Dying From Heart Failure?
An athletic 20 year man is playing basketball and suddenly collapses on the court and dies. On a hot July day, a young and vibrant college football player suddenly makes a great tackle and never [...]
CONTACT US
Send A Message To Leading Edge Medical
If you have any questions, concerns, or comments regarding Leading Edge Medical, please fill out the short contact form below.